If 2022 was a sparse 12 months for US Meals and Drug Administration approvals of recent molecular entities, 2023 was a return to the tempo the company had achieved from 2017 by way of 2021. By mid-September, the FDA had already surpassed the 37 approvals it had made in all of 2022 and by mid-November reached 50 approvals.
Along with approving firsts in prescribed drugs and vaccines, the company permitted over-the-counter variations of a contraception capsule and naloxone, used for reversing opioid overdoses, for the primary time. Additionally making headlines this 12 months, FDA advisory panels scrutinized an ingredient in widespread over-the-counter chilly medicines and a trial of a first-in-class most cancers drug.
Leqembi was permitted to deal with Alzheimer’s illness
Credit score: Eisai
Eisai and Biogen’s Leqembi (lecanemab) turned the second monoclonal antibody drug to obtain accelerated FDA approval to deal with Alzheimer’s illness on Jan. 6. It then acquired full approval from the company—a primary for this class of Alzheimer’s remedy—on July 6. In 2021, the FDA granted accelerated approval to Biogen and Eisai’s monoclonal antibody Aduhelm (aducanumab), however the choice was controversial; many medical doctors and scientists argued that medical trials of Aduhelm confirmed no profit to sufferers. Each medicine clear amyloid-β deposits within the brains of individuals with Alzheimer’s illness. However not like Aduhelm, Leqembi slowed cognitive decline, albeit modestly, in medical trials of individuals with early-stage Alzheimer’s. The FDA is anticipated to determine on approval for one more monoclonal antibody remedy for Alzheimer’s, Eli Lilly and Firm’s donanemab, by the top of the 12 months.
Zurzuvae bought the nod for the remedy of postpartum despair however not main depressive dysfunction
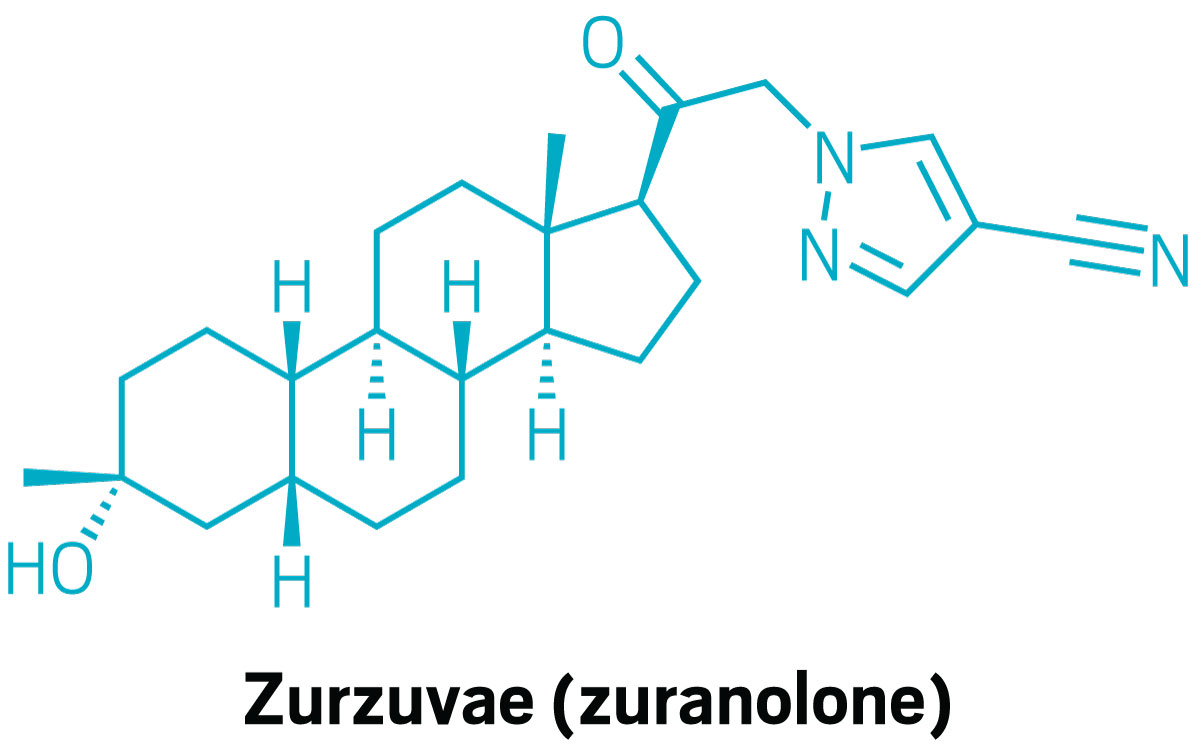
The FDA granted approval to Sage Therapeutics and Biogen’s Zurzuvae (zuranolone) as the primary oral remedy for postpartum despair on Aug. 4. Beforehand permitted therapies for postpartum despair must be injected. Zurzuvae is a neuroactive steroid that modulates γ-aminobutyric acid (GABA) receptors within the central nervous system, thereby calming nerve exercise. However the FDA rejected the drug as a remedy for main depressive dysfunction. That break up choice was dangerous information for each companies, which noticed their share costs take a knock. Sage subsequently introduced a reorganization that included axing 40% of its workforce.
RSV immunizations turned obtainable
Credit score: Nationwide Institute of Allergy and Infectious Illnesses/Science Supply
An artist’s depiction of respiratory syncytial virus
On Could 3, the FDA permitted GSK’s Arexvy, the primary vaccine for respiratory syncytial virus (RSV) to be used in individuals aged 60 and older. Earlier than Could was over, the company additionally permitted Pfizer’s Abrysvo, a second RSV vaccine for a similar age group. On Aug. 21, Abrysvo additionally acquired the FDA’s approval to be used in pregnant people between 32 and 36 weeks’ gestation to forestall RSV in infants from start to six months of age. Each vaccines use an RSV floor protein referred to as the fusion glycoprotein to provoke an immune response from the physique. The company green-lit a unique kind of RSV immunization on July 17 when it permitted AstraZeneca and Sanofi’s Beyfortus (nirsevimab), a monoclonal antibody that may be given to infants and youngsters as much as 24 months in age. Moderna submitted an software to the FDA for its messenger RNA–based mostly RSV vaccine in July. Bavarian Nordic’s vaccine, which featured a reside virus adorned with RSV proteins, failed to satisfy all its main finish factors, prompting the corporate to discontinue its RSV program.
A contraception capsule and Narcan turned obtainable over-the-counter

Credit score: Emergent BioSolutions/Perrigo
The FDA first permitted norgestrel as a prescription oral contraceptive in 1973. Fifty years later, on July 13, the company permitted Perrigo’s Opill as a model of this contraception capsule that doesn’t require a prescription.
In one other notable over-the-counter approval, the FDA made Narcan (naloxone), Emergent BioSolutions’ nasal spray for reversing opioid overdoses, obtainable to shoppers with no prescription on March 29. Beforehand, advocates had managed to work across the drug’s prescription-only standing through a patchwork of state legal guidelines, standing orders, and collaborative follow agreements amongst pharmacists.
Lumakras research was referred to as into query
A Part 3 trial that was meant to substantiate the information that gained Amgen’s Lumakras (sotorasib) expedited approval didn’t cross muster with a panel of FDA advisers. In October, the panel voted 10–2 that the CodeBreaK 200 trial outcomes couldn’t be reliably interpreted.
In 2021, the FDA granted expedited approval to Lumakras to deal with individuals who have superior non-small-cell lung most cancers pushed by theKRAS G12C mutation and had beforehand taken one other chemotherapy. Lumakras was the primary drug to inhibit KRas, a notoriously difficult goal.
The FDA required additional information to transform its accelerated approval into full drug approval. The research that was supposed to offer these information, CodeBreaK 200, confirmed that progression-free survival was a few month longer for individuals who took Lumakras in contrast with individuals who took docetaxel, a typical second-line remedy for any such most cancers. The research discovered no distinction in total survival.
However the FDA advisers questioned CodeBreaK 200’s outcomes. They cited a excessive dropout fee amongst sufferers within the docetaxel arm of the research, which can have launched bias. The advisers have been additionally involved as a result of unbiased evaluations of tumor imaging scans differed considerably from these carried out by the research’s investigators.
The advisory panel’s conclusion doesn’t imply that Lumakras shall be withdrawn from the market, however additional research could also be wanted. The FDA will make its choice on Lumakras’s full approval later this month.
Phenylephrine isn’t an efficient oral decongestant, in response to an FDA panel
In 2022, individuals within the US spent an estimated $1.8 billion on over-the-counter drugs and liquids containing phenylephrine, equivalent to DayQuil and Sudafed PE, in response to producer gross sales information compiled by the FDA. However this September, an FDA advisory panel agreed in a unanimous vote that the ingredient doesn’t work as a decongestant when taken orally.

Credit score: Bethany Halford/C&EN
Drug retailer cabinets are filled with oral cough and chilly medicines that include phenylephrine.
Phenylephrine, which has been used medically for the reason that Nineteen Thirties, constricts blood vessels. The FDA has deemed it typically acknowledged as secure and efficient, or GRASE within the company’s parlance, for over-the-counter medicines since 1976. For many years, although, scientists have argued that the FDA’s choice was based mostly on flawed information. They are saying that when taken orally, phenylephrine is shortly metabolized within the intestine to inactive molecules and by no means makes its manner into the bloodstream. Nasal sprays that include phenylephrine are nonetheless thought-about efficient at stopping runny noses.
The drug got here into widespread use in oral formulations after the US federal authorities enacted the Fight Methamphetamine Epidemic Act of 2005. That laws required pharmacies to maintain chilly cures with a unique decongestant—pseudoephedrine, which may be simply transformed to methamphetamine —behind the counter, and it required shoppers to point out identification to buy them. Drugmakers used phenylephrine to fill the general public’s demand for chilly medicines.
The FDA panel reviewed the previous information together with the outcomes of more moderen medical trials that confirmed that oral phenylephrine didn’t work, even at doses greater than these advisable on the packaging. The FDA is contemplating the panel’s suggestion and can make the ultimate choice about whether or not phenylephrine ought to preserve its GRASE designation, however the course of may take greater than a 12 months.